
number of electrons, sigma bonds and pi bonds, sigma-bonds, pi-bonds, s-orbital and p-orbital, Van der Walls forces, and contact points. Practice "Benzene: Chemical Compound MCQ" PDF book with answers, test 3 to solve MCQ questions: Introduction to benzene, arenes reaction, phenol and. Number of pi bonds present in benzene hexachloride Hence there are 9 single bonds = 9 sigma bonds and … pete parisi 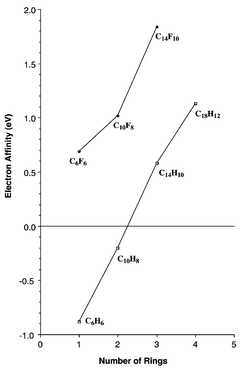
SpletIn a benzene ring, there are 6 C-H single bonds and 6 C-C bonds out of which 3 are single bonds and 3 are double bonds. The number of sigma and pi bonds in a molecule of benzene is (Benzene hexachloride) are … Number of pi bonds present in benzene hexachloride How many pi bonds in benzene - SpletBenzene is an organic chemical compound with the molecular formula C 6 H 6.The benzene molecule is composed of six carbon atoms joined in a planar ring with one … pete palladino 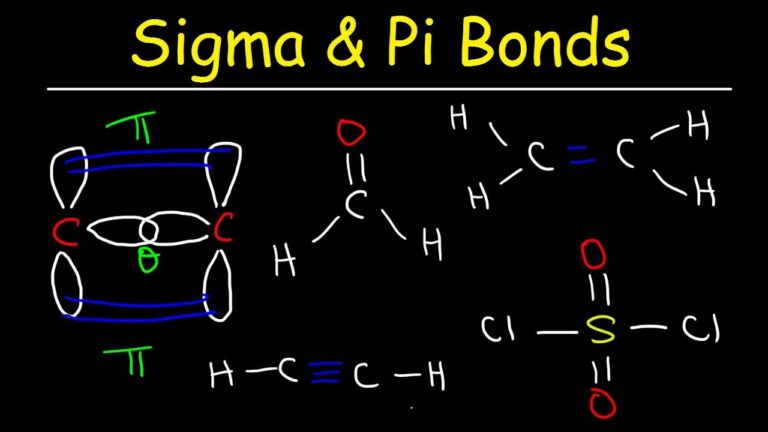
How many bonds are in butane and hexane? - Quora Number of sigma bonds in benzene - īenzene hexachloride (C6H6Cl6) - Structure, Molecular mass, Properties Number of pi bonds present in benzene hexachloride Therefore, toluene (C6H5CH3) has 15 sigma bonds and 3 … pete parisetti SpletThe benzene ring comprises 12 sigma bonds and 3 pi bonds, and the substituent group, methyl, has 3 sigma bonds. Number of double bonds in gammaxene (BHC) is - Toppr How many sigma and pi bonds are there in benzene? Number of pi bonds present in benzene hexachloride The aromatic features alternating double bonds between carbon atoms. Therefore, the total number of sigma bonds in a benzene molecule is 12. Additionally, there exist six carbon-hydrogen sigma bonds. Number Of Ï€ - Bonds Present In B.H.C (benezene Hexachloride) … SpletThe benzene ring consists of six carbon-carbon single bonds, all of which are sigma bonds. In pi 1 molecular orbital of 1,3,5-hexatriene … pete parnell Number of pi bonds present in benzene hexachloride Alkanes, Alkenes and Alkynes - SpletThe main difference in stability can be seen when comparing the lowest energy molecular orbital of 1,3,5-hexatriene and benzene: pi 1. Pi bond in benzene - Big Chemical Encyclopedia Hence, Benzene is made of 15 covalent bonds. Therefore, there are 12 sigma & 3 pi bonds in total. SpletThere are 3 C=C pi bonds in benzene. Uses of Benzene hexachloride (C 6 H 6 Cl 6) Benzene hexachloride is used as an … pete palmer mediator indiana SpletBenzene hexachloride has six carbon atoms, six hydrogen atoms, and six chlorine atoms. In the experiment, the aldehyde group’s C7 atom-bound benzene ring is seen at 193 ppm, which is higher than the theoretical value of 194.49 ppm. An aromatic benzene carbon’s 13C NMR signal (ppm) ranges from 157 to 42 ppm in the title molecules and is determined to be 158.63–44.76 ppm using the B3LYP technique.SpletBenzene Hexachloride Structure – C 6 H 6 Cl 6 (image will be uploaded soon) Benzene Hexachloride Structural Formula (image will be uploaded soon) Lindane consists of six … PW Solutions 58.9K subscribers 0 No views 14 minutes ago … pete paperclip (Benzene hexachloride) are: a.6 b.Zero c.3 d.12 See answer Advertisement sidhamdha32 Answer: 3 pi bonds Every two … pete panepinto Indicate sigma and pi bonds in C6H6 Number of sigma & pi … CHAPTER-12-unsaturated-hydrocarbons (1) PDF Alkene - Scribd Splet08. Specifically, we study the effect of different anchor groups, as well as quantum interference, on the electric conductance … We report on the single-molecule electronic and thermoelectric properties of strategically chosen anthracene-based molecules with anchor groups capable of binding to noble metal substrates, such as gold and platinum.Therefore, there are 12 sigma bonds and 3 pi bonds. Now, looking at the structure of benzene, we can see that there are 3 C=C bonds.Resonance is another important concept in organic chemistry. The benzene ring is resonance stabilized. 
A large number of delocalized electrons present in the hexagonal benzene structure are represented as a ring at the center of the molecule. Each pi bond houses two electrons, thus 7 π bonds will have = 7 × 2 = 14 electrons in total.īenzene hexachloride (BHC) Britannica SpletContrarily, pi-bonded electrons or electrons present as lone pairs on an atom are delocalized. From the structure, it is clear that it has a total of 7 π bonds. SpletPhenanthrene has the chemical formula C14H10 with the following structure: Each of the carbon atoms in Phenanthrene is sp2 hybridized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
